Fructose and glucose are very similar from a chemical standpoint, and both are six carbon sugars with the same molecular weight. However, what distinguishes them is how they are metabolized. Fructose can be metabolized identically as glucose by an enzyme called hexokinase, but fructose is preferentially metabolized by an enzyme called fructokinase (also known as ketohexokinase, or KHK). This is the first enzyme in fructose metabolism and it uses ATP to phosphorylate fructose to generate fructose-1-phosphate (Figure).
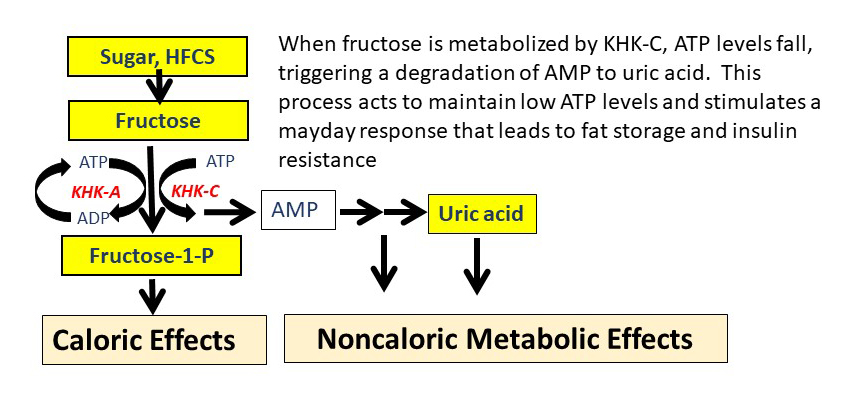
There are two different KHK enzymes. One isoform is KHK-C, and this is the main enzyme that metabolizes fructose. It is present in the liver, intestine, kidney, islets of the pancreas, and in the brain. It can also be induced under conditions of stress, such as in the heart following a heart attack. The other enzyme, KHK-A is more ubiquitous, but metabolizes fructose much more slowly compared to KHK-C.
One of the key differences in KHK-A and KHK-C is that KHK-C will phosphorylate fructose so quickly that it can cause local ATP depletion. In contrast, KHK-A does not lower ATP. The enzymes that metabolize glucose also do not lower ATP levels, for while glucokinase and hexokinase also use ATP, they have a regulatory feedback system that turns off the process if ATP levels start to fall.
Our group has been able to show that KHK-C is the master regulator activating the fat switch, and that this is linked with its ability to reduce ATP levels. We showed this by giving fructose to mice that had no KHK, in which we could completely block the development of metabolic syndrome even if the animals were forced to eat fructose. In contrast, if we only blocked KHK-A (using KHK-A knockout mice), the animals actually developed worse obesity and metabolic syndrome, as more of the fructose was metabolized by KHK-C.
Thus, Fructokinase-C is the key enzyme responsible for how fructose causes metabolic syndrome. As such, large pharma and other companies are trying to develop inhibitors to block KHK-C activation.